- Hits: 3831
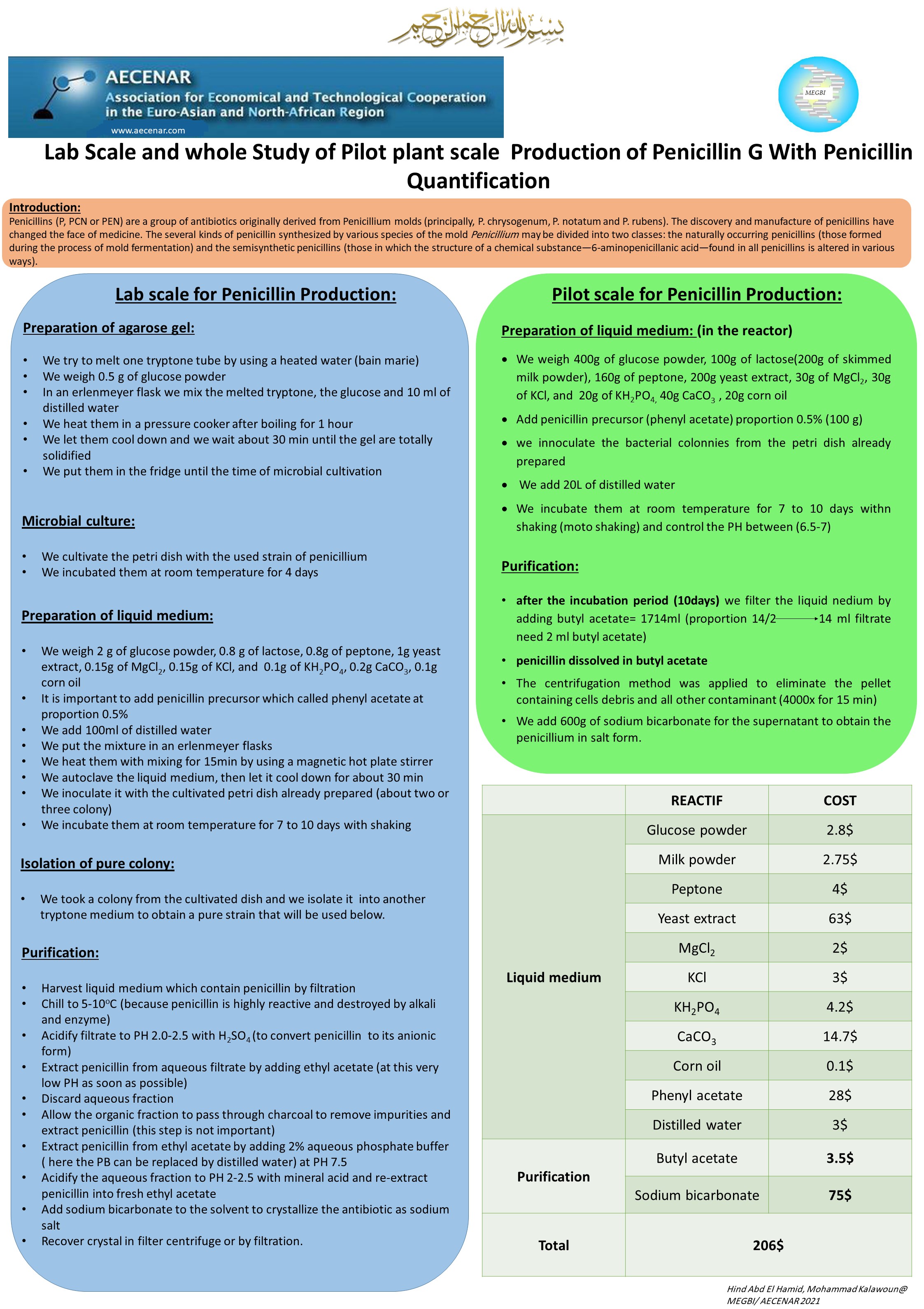
Lab Scale Penicillin Production
21.10.2022:
19.10.2022:
Table 1: Constitutes of penicillin G fermentation broth(1)
Table 2: Parameters monitored in penicillin fermentation(1)
Penicillin purification and crystallization(2):
- First the filtrate is treated with H2SO4 solution to reach a pH between 1.5 and 2.2(Here the penicillin is presented ionized into the solution).
- Then an organic solvent must be added (Butyl acetate)(Here the ionized penicillin is present in the organic phase)
- To extract the penicillin from the organic phase an aqueous phase containing sodium or potassium ion must be added with the adding of NaOH to reach a pH from about 6 to 8
- Here the crystal of penicillin must be formed and then filtrated
Crystallization(3):
- From filtration, the penicillin rich solution is cooled to 5°C. As penicillin G only has a half-life 15 minutes at pH 2 at 20°C, this helps reduce enzyme and chemical degradation during the solvent extraction step.
- Crystals are highly organised inert matters. If grown without external interference, they grow in polyhedral shapes and exhibit many degrees of symmetry. Penicillin G is an odourless, colourless or white crystal, or crystalline powder. Crystallisation is essentially a polishing step that yields a highly pure product. It is done through phase separation from a liquid to a solid. To begin crystallisation, we must first have a supersaturated solution. Supersaturation refers to a state in which there are more dissolved solids in the solvent than can ordinarily be accommodated at that temperature at equilibrium. Supersaturation can be achieved usually by cooling, drowning, solvent evaporation, or by chemical reaction. Since the solubility of penicillin in its aqueous solution decreases with decreasing temperature, as the solution cools, its saturation increases until it reaches supersaturation and crystallization begins.
18.10.2022
.Testing the sensibility of staphylococcus aureus strain against penicillin g
- preparation of stock solution of penicillin g (1MUI=1000000 unite internationale)
- Achievement of serial dilution to reach a concentration of 1000 UI
- applied of this final concentration into the surface of standard agar
- Strain culture of staph aureus with the presence of control plate(without penicillin)
- Incubation for 20-24 h at 37oc
- The results is mentionned below:
1- Experiment 2: Penicillin production
- The same steps already mentioned in a last report are followed(10).
Penicillin purification:
To purify the produced penicillin we had pursued different three protocol.
The first (P1) without using charcoal (just: filtration-ethyl acetate-centrifugation- sodium bicarbonate)
The second by using charcoal and V/V from the ethyl acetate
The third by using charcoal and V[14:2] from the ethyl acetate( for each 14 ml of the filtrate use 2 ml of ethyl acetate)
Then we have tested the filtrate when we had obtain a lot of contaminant.
For this reason the protocol was retried with the autoclaving of the liquid medium before adding the penicillium colony. Then the quantification will be tried.
2- Corrected protocol for Penicillin production :
2.1 Purification of the isolate:
We took a colony from the cultivated dish and we recultivate into another tryptone medium to obtain a pure strain that will be used below.
2.2 Preparation of liquid medium
- We weigh 2 g of glucose powder, 0.8 g of lactose, 0.8g of peptone, 1g yeast extract, 0.15g of MgCl2, 0.15g of KCl, and 1g of KH2PO4, 0.2g CaCO3, 0.1g corn oil
- It is important to add penicillin precursor which called phenyl acetate at proportion 0.5%
- We add 100ml of distilled water
- We put the mixture in an erlenmeyer flasks
- We heat them with mixing for 15min by using a magnetic hot plate stirrer
- We autoclave the liquid medium, then let it cool down for about 30 min
- We inoculate it with the cultivated petri dish already prepared (about two or three colony)
- We incubate them at room temperature for 7 to 10 days with shaking
- Harvest liquid medium which contain penicillin by filtration
- Chill to 5-10oC (because penicillin is highly reactive and destroyed by alkali and enzyme)
- Acidify filtrate to PH 2.0-2.5 with H2SO4 (to convert penicillin to its anionic form)
- Extract penicillin from aqueous filtrate by adding ethyl acetate (at this very low ph as soon as possible)
- Discard aqueous fraction
- Allow the organic fraction to pass through charcoal to remove impurities and extract penicillin (this step is not important)
- Extract penicillin from ethyl acetate by adding 2% aqueous phosphate buffer ( here the PB can be replaced by distilled wter) at PH 7.5
- Acidify the aqueous fraction to PH 2-2.5 with mineral acid and re-extract penicillin into fresh ethyl acetate
- Add sodium bicarbonate to the solvent to christallise the antibiotic as sodium salt
- Recover crystal in filter centrifuge or by filtration.
Pilot plant penicillin production.docx
Bioreactor and antibiotic production pilot plant.docx

3-New corrected for the proposed protocol: (22/8/2022)
This time we used the broth medium for penicillium maintaining and growth and continue same steps
Broth description: Broth cultures are a method of growing bacteria in a liquid growth medium. They're used to grow and maintain cultures for a laboratory. Different bacteria may grow differently in broth cultures. They are: Aerotolerant anaerobic bacteria evenly dispersed throughout the culture.

broth medium after 4 days of incubation with penicillum trisogome
-And we preapred new liquid medium to test and compare the penicillin production with the standard medium used before


the new medium proposed (I&II) and the stanard medium ready to be incubated from 7 to 10 days
The next step is to filtrate the 2 medium prepared (31/7/2022).

filtration (whatman paper) the medium I & II
And the last step is sensitivity test on the filtrate (1/8/2022).

the surface of the gel is coverd by the mediums filtrate and staphylococus aureus
